
In 1946, electrocoagulation was first applied to treat drinking water in the US 3. And at the end of this paper, the characteristics of new combination methods, such as photocatalysis, nanofiltration, and the use of biochemical method, are discussed. The earliest application of electrochemical technology in water treatment was reported in the United Kingdom in 1889, a century after the first use of electricity to treat water. Vukovi, Modern water treatment by electrochemical oxidation a review, Tmt, 2013, 10-11. This paper reviews recent developments in electrode materials for electrochemical degradation of halogenated organics. Much research has therefore been devoted to developing appropriate electrode materials for practical applications. 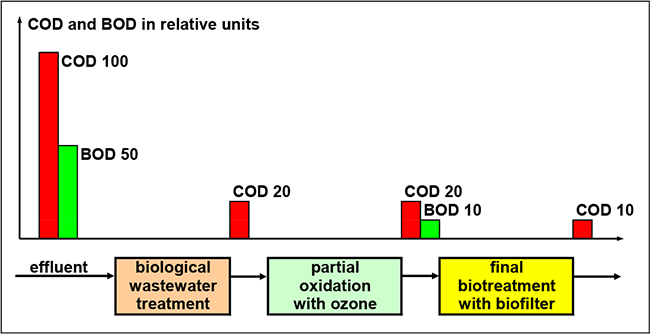
The electrode material is crucial to the degradation efficiency of an electrochemical process. Electrochemical techniques are classified according to main reaction pathways: (i) electrochemical reduction, in which cleavage of C-X (X = F, Cl, Br, I) bonds to release halide ions and produce non-halogenated and non-toxic organics and (ii) electrochemical oxidation, in which halogenated organics are degraded by electrogenerated oxidants. Electrochemical methods are attractive and promising techniques for wastewater treatment and have been developed for degradation of halogenated organic pollutants under mild conditions. 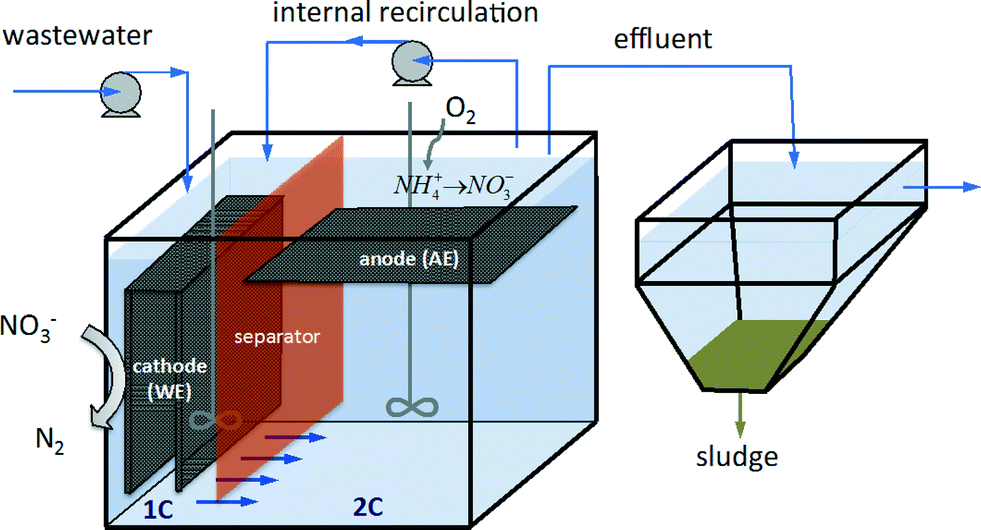
Halogenated organics are widely used in modern industry, agriculture, and medicine, and their large-scale emissions have led to soil and water pollution.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
